Figure 1. T-cell specificity profiling. (Hadrup, 2017)
T Cell Specificity Assay
ProMab Biotechnologies has developed a comprehensive system for CAR-T cell research and development. To optimize TCR research, our team has designed a strategy to offer T cell specificity assays to meet your project's needs.
It is well known that the recognition of short peptide sequences presented on MHC class I by the T cell receptors (TCRs) of T lymphocytes is essential for the defenses against malignant cells and pathogens. In adoptive cell transfer (ACT) therapy, the functional activity of large populations of T cells can be redirected against defined targets by antigen-specific TCRs engineering.
ProMab provides high-quality in vitro assays to test the specificity of engineered T cells for interested antigens, alongside our platform for CAR-T and TCR modified T cell research and development.
ProMab Biotechnologies Custom T Cell Specificity Assays
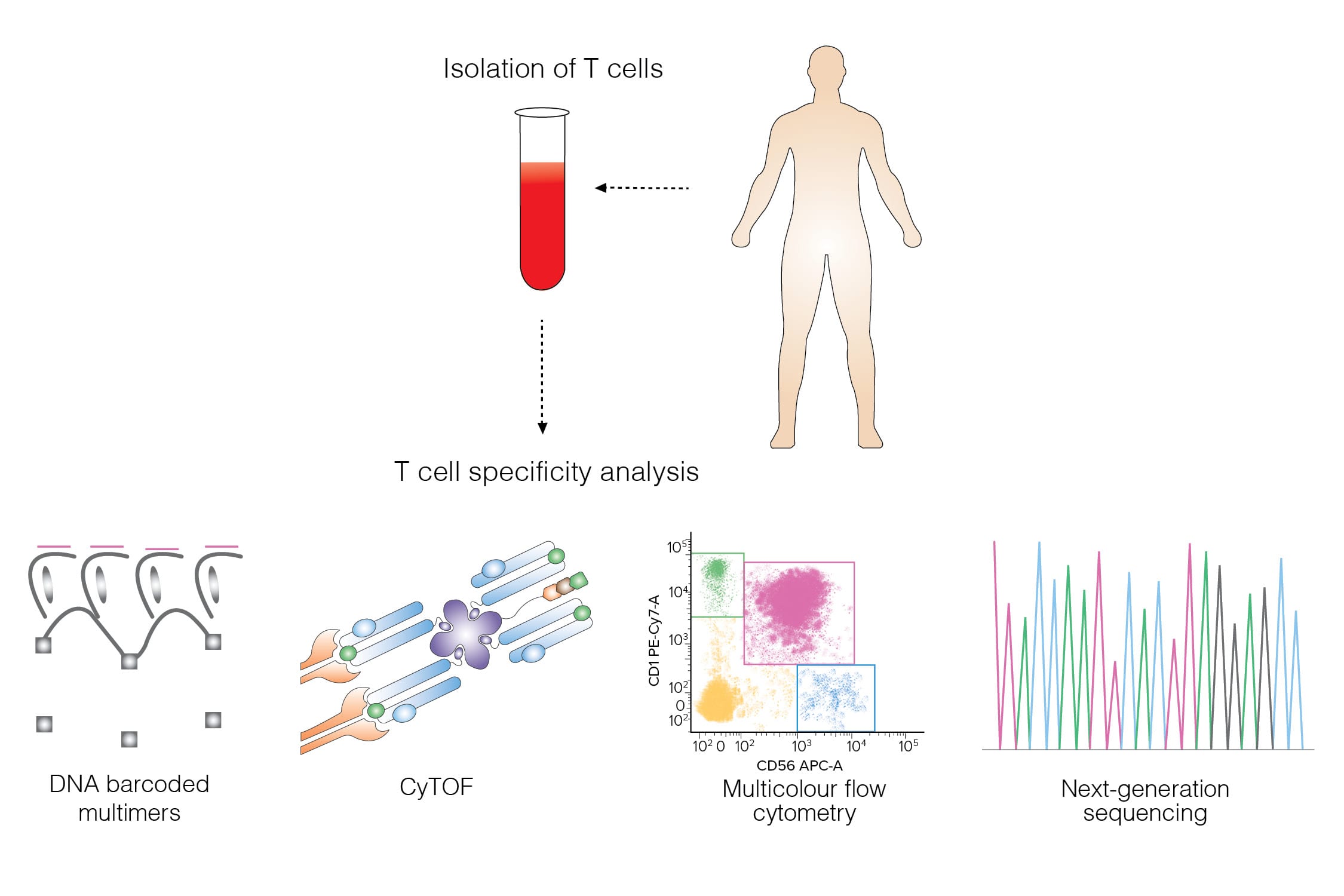
Understanding T cell recognition patterns and T cell specificity in health and disease plays a critical role in the therapeutic efficacy of cell and gene therapy-based IO drugs development.
We have developed a variety of in vitro T cell specificity assays to analyze the T-cell epitopes and functions, for example flow cytometry, next-generation sequencing, and MHC multimer technology for probing antigen specificity.
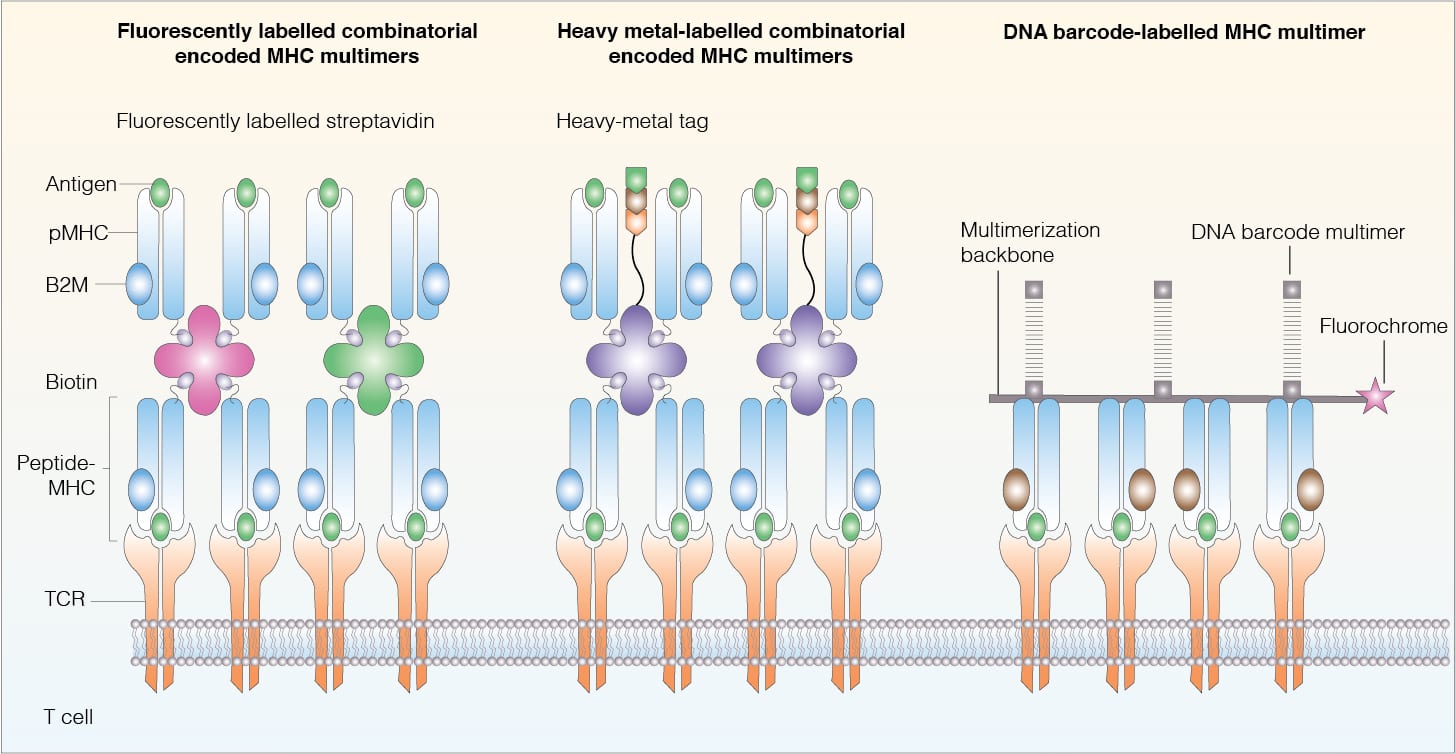
There are three multiplex MHC multimer technologies have been applied in T cell specificity assays, namely heavy-metal-tagged combinatorial encoded MHC multimers, fluorescently labeled combinatorial encoded MHC multimers and DNA-barcode-labelled MHC multimers.
Contact ProMab today to discuss T cell specificity assay services and more!
Other Components of ProMab's Cell Based Assay Platform:
Reference
Hadrup, S. and Newell, E. (2017). Determining T-cell specificity to understand and treat disease. Nature Biomedical Engineering. 1(10), pp.784-795.




